

Even as recently as the late 1990s, patients did not have to choose between types of artificial discs. There was a lumbar artificial disc, and then there was a cervical artificial disc. Today, patients and their spine surgeons can choose from many different types of artificial discs. This provides freedom, but it also makes things a bit more complicated. Fortunately, your spine surgeon can help you choose the best kind of artificial disc replacement for your specific needs.
Cervical Artificial Discs


The BRYAN® Cervical Disc
Off Market
The BRYAN® Cervical Disc is comprised of two titanium alloy shells that surround a polycarbonate urethane nucleus. The titanium alloy shells are roughened to foster bone growth within the surface pores. Unlike newer discs that are a bit more forgiving, the BRYAN® Cervical Disc requires extremely precise placement for it to function properly. This steep technical requirement and somewhat outdated design means that BRYAN® Cervical Disc is rarely implanted today. However, it delivered good results for patients who received them in the late 2000s and early 2010s. This disc is currently off the market.


PCM® Cervical Disc
Off Market
The PCM® Cervical Disc is comprised of two cobalt-chromium metal alloy endplates; however, sitting on top of and fully integrated with the lower endplate is a hard plastic spacer. The sides of the disc that face bone are covered with titanium and calcium phosphate, the latter being a bone-like substance. While the PCM® Cervical Disc was initially approved by the FDA in 2012, it is no longer available. This disc is currently off the market.


Prodisc-C® Cervical Disc
The Prodisc-C® Cervical Total Disc Replacement consists of three parts: two cobalt-chromium-molybdenum metal alloy endplates surrounding a polyethylene (ultra-high molecular weight plastic) inlay. The three components lock into place, forming a single, functional device with good range of motion. The upper and lower metal surfaces are sprayed with titanium plasma. This titanium plasma spray provides a surface for the vertebral bones to attach and integrate.


PRESTIGE® Cervical Disc
Unlike the Prodisc-C® device, the PRESTIGE® Cervical Disc does not contain cobalt or chromium. Instead, the current PRESTIGE LP® Cervical Disc is comprised of a titanium ceramic composite and titanium carbide. The titanium ceramic composite has an excellent wear rate when subjected to motion tests. The two-piece configuration lacks a mobile core, which in other devices could conceivably migrate out of the intervertebral space. The PRESTIGE LP® Cervical Disc is sturdy while allowing outstanding range of motion. Clinical trials results with the PRESTIGE® Cervical Disc device have been superb.


M6-C™ Artificial Cervical Disc
The M6-C™ Artificial Cervical Disc has four components but is integrated into a single device. Much like the natural disc, the M6-C™ Artificial Cervical Disc contains a tough outer annulus and a softer, inner nucleus. The artificial annulus is made of a polyethylene fiber, and the artificial nucleus is made of a viscoelastic (polycarbonate urethane) polymer. The artificial nucleus and annulus are surrounded above and below by titanium endplates. The titanium plasma spray-coated, titanium endplates have fixation keels (raised ridges) that press in to hold to the spinal bones. The M6-C™ Artificial Cervical Disc provides good stability and range of motion in the cervical spine.

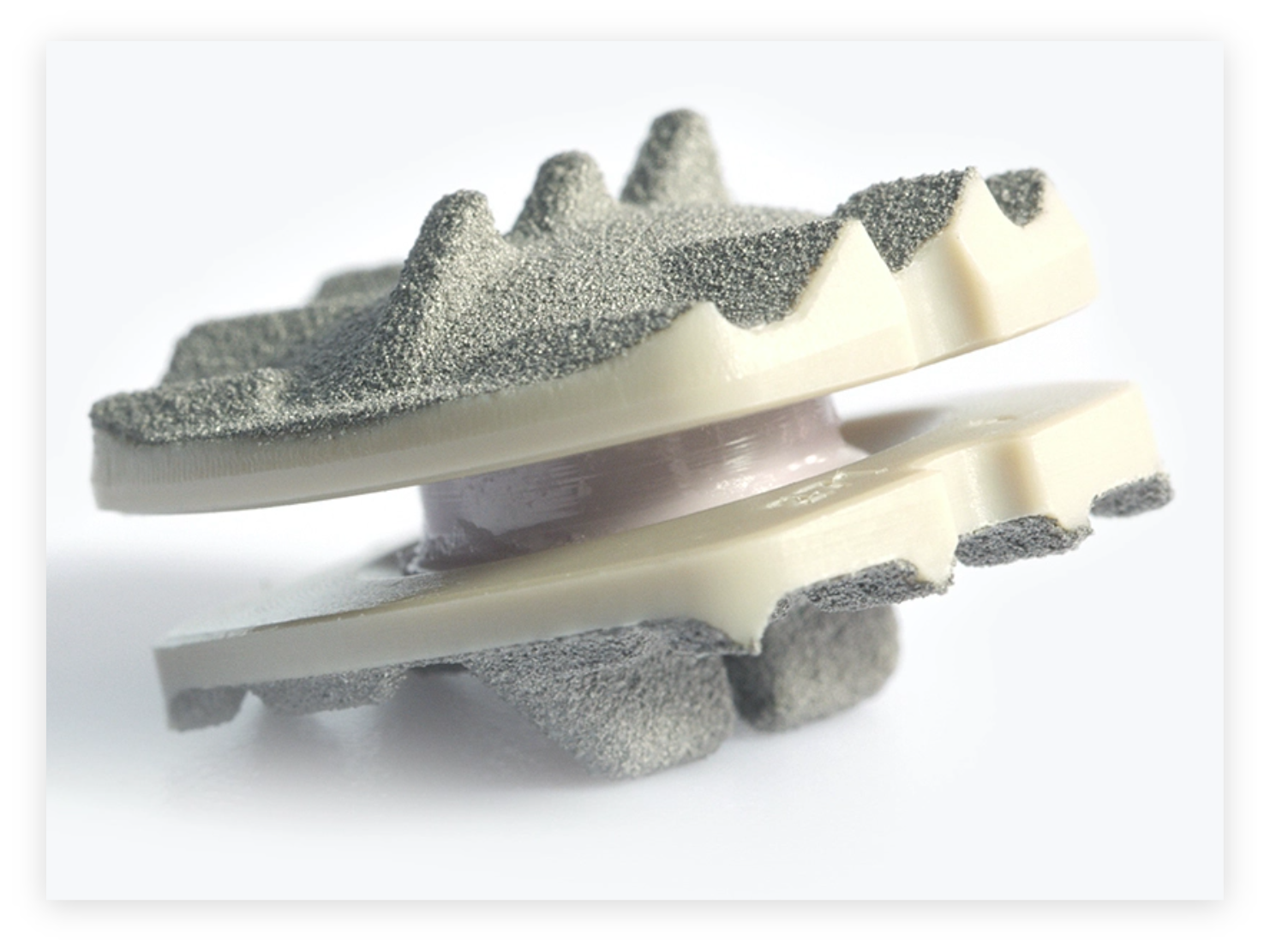
Simplify® Cervical Artificial Disc
The Simplify® Cervical Disc offers the lowest disc heights of any of the major cervical disc manufacturers. In practical terms, this allows your spine surgeon to place an artificial disc that is just the right size for the intervertebral space. Another interesting feature of the disc is that it is comprised solely of MRI-friendly materials, namely hard plastic polymer endplates and a ceramic core that is fortified with zirconium and aluminum. The endplates are sprayed with titanium plasma and contain low-rise keels that help the device integrate with vertebral bone.


Mobi-C® Cervical Disc
Mobi-C® Cervical Disc – One unique feature of the Mobi-C® Cervical Disc is that it contains a mobile, polyethylene bearing that adjusts as the cervical spine moves. This bearing rests between two cobalt-chromium-molybdenum metal alloy endplates, which are in turn coated in titanium and hydroxyapatite (a bone-like material). The central bearing provides exceptional joint mobility but carries a theoretical risk of displacement. The addition of hydroxyapatite may increase the speed and stability of integration of artificial device with bone.


SECURE-C® Cervical Disc
Like other devices mentioned above, SECURE-C® Cervical Disc is comprised of two cobalt-chromium-molybdenum metal alloy endplates and a polyethylene (hard plastic) core. These three separate pieces provide substantial joint flexibility and, consequently, spinal motion. In mathematical terms, the SECURE-C® Cervical Disc permits 15 degrees of motion as the neck flexes and extends and 10 degrees of motion as the head moves from side to side. The disc also has unconstrained axial rotation, which means the artificial cervical disc allows the patient’s head to turn as freely as any other natural disc. Serrated keels and a titanium plasma spray facilitate bone integration. Patient satisfaction rates after SECURE-C® Cervical Disc placement were high.
Other Artificial Cervical Discs That Are Retired, Active, Or In Development
- PRODISC C Nova
- PRODISC C Vivo
- activ C
- Aditus Cervical disc prosthesis
- Almas
- Aramis
- Baguera C
- Biolign Disc
- c|spinedisc
- d-flex Titanium
- d-motion
- d-active
- Crea Spine Cervical disc prosthesis
- Discocerv
- DYNALIS-C®
- ESP Cervical Disc
- Freedom®
- GRANVIA® C
- Kineflex Cervical Disc
- LorX® Cervical Disc
- Prosthesis
- MOVE-C®
- NEOphytos
- PAN Cervical Disc Prosthesis
- Procoral Artificial Disk
- ROTAIO
- Synergy Cervical Disc
- Triadyme C Cervical Disc
- Viking Spine Ceramic
- Viking Spine Titanium
Lumbar Artificial Discs


CHARITÉ® Artificial Disc
The CHARITÉ® Artificial Disc is the pioneering artificial disc because it was the first total disc replacement device to be approved for use. It is comprised of a single piece that is inserted into the intervertebral space. The original CHARITÉ® Artificial Disc was updated two times, resulting in a Type III version, however it is no longer available. The INMOTION® lumbar artificial disc by DePuy Spine is a modified version of the CHARITÉ® Artificial Disc, but it too is not available for use in the United States.


Prodisc-L® Lumbar Disc
The Prodisc-L® Total Lumbar Disc Replacement uses a ball-and-socket design similar to the configuration used in hip replacements for over three decades. The device has a central keel and lateral spikes on its titanium plasma-sprayed, cobalt-chromium alloy endplates. The “ball” is this system is an ultra-high molecular weight polyethylene inlaid core. Approved in 2005 for one-level disease and in 2020 for two-level disease, the Prodisc-L® Total Lumbar Disc Replacement system is one of the oldest, consistently used device platforms on the market.


activ L (discontinued)
Like the Prodisc-L®, the activ L artificial disc is has two titanium-coated cobalt-chromium metal alloy endplates and an ultra-high molecular weight polyethylene core; however, the core is mobile in the activ L device. Unlike the Prodisc-L®, the activ L artificial core is flattened on one side—the curved side provides range of motion while the flattened side provides stability. Similar to the Simplify® device for cervical discs, the activ L artificial lumbar disc has a very low overall height, which means it can replace natural discs with only limited space between vertebrae.
Other Artificial Lumbar Discs That Are Retired, Active, Or In Development
- BAGUERA® L
- Freedom® Lumbar Disc
- Flexicore
- Kineflex
- LP-ESP disc prosthesis
- Mobidisc® L Lumbar Disc Prosthesis
- M6-L artificial lumbar disc
- ORBIT™-R
Your Spine Surgeon
Will Help You Decide
What Artificial Disc is
Right for You.
The number of artificial discs—especially for the cervical spine—is large and growing. While this can mean choosing the best artificial disc type is daunting for patients, the wide variety of design options and construction materials means that patients have extensive freedom to choose the product that is right for them. Fortunately, experienced spine surgeons can narrow down the possible list of artificial discs by listening to your detailed history, carefully reviewing your imaging studies, and providing you with clinical insights gained from dozens of artificial disc replacement procedures.
Ready to reclaim your life? Get in touch with Dr. Lanman Today.
FOLLOW US ON SOCIAL MEDIA | @ADRSPINE



